Volume 21, Number 2—February 2015
CME ACTIVITY - Research
pH Level as a Marker for Predicting Death among Patients with Vibrio vulnificus Infection, South Korea, 2000–2011
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; (4) view/print certificate.
Release date: January 16, 2015; Expiration date: January 16, 2016
Learning Objectives
Upon completion of this activity, participants will be able to:
• Describe characteristics of patients infected with Vibrio vulnificus
• Assess the outcome of infection with V. vulnificus
• Distinguish significant prognostic factors among patients infected with V. vulnificus
• Identify the cutoff value for arterial pH with the best discretion for the prognosis of V. vulnificus infection
CME Editor
Claudia Chesley, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Claudia Chesley has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Clinical Professor of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed the following financial relationships: served as an advisor or consultant for McNeil Pharmaceuticals.
Authors
Disclosures: Na Ra Yun, MD; Dong-Min Kim, MD, PhD; Jun Lee, MD; and Mi Ah Han, MD, PhD, have disclosed no relevant financial relationships.
Abstract
Vibrio vulnificus infection can progress to necrotizing fasciitis and death. To improve the likelihood of patient survival, an early prognosis of patient outcome is clinically important for emergency/trauma department doctors. To identify an accurate and simple predictor for death among V. vulnificus–infected persons, we reviewed clinical data for 34 patients at a hospital in South Korea during 2000–2011; of the patients, 16 (47%) died and 18 (53%) survived. For nonsurvivors, median time from hospital admission to death was 15 h (range 4–70). For predicting death, the areas under the receiver operating characteristic curves of the Acute Physiology and Chronic Health Evaluation (APACHE) II score and initial pH were 0.746 and 0.972, respectively (p = 0.005). An optimal cutoff pH of <7.35 had a sensitivity of 100% and specificity of 83%. Compared with the APACHE II score, the initial arterial blood pH level in V. vulnificus-infected patients was a more accurate predictive marker for death.
Vibrio vulnificus is a motile, halophilic, rod-shaped gram-negative pathogen that lives in estuarine environments (1). During the last decade, the prevalence of V. vulnificus infection has increased worldwide (2–4). In the United States, the annual number of V. vulnificus infections reported to the 10-state Foodborne Disease Active Surveillance Network (http://www.cdc.gov/foodnet/) increased from 0.01 cases/100,000 persons in 1996 to 0.05 cases/100,000 persons in 2010 (5).
V. vulnificus infections, which are mainly characterized by skin and soft-tissue infections or septicemia, can develop a fulminant course (1,6). The severe form of V. vulnificus soft-tissue infection, necrotizing fasciitis, often has adverse outcomes, including death. In such cases, death can occur within 48 h of hospital admission, especially if the infection is associated with the development of sepsis or septic shock, which increases the case-fatality rate to 26%–71% (7–11). For cases of V. vulnificus infection caused by the consumption of contaminated shellfish, the death rate is ≈53%, and the rate is higher (67%) among patients with liver disease (12). The death rate may increase to 100% in patients with septicemia if treatment is delayed for 72 h after symptom onset (13).
Surgery, including fasciotomy, debridement, and limb amputation, early in V. vulnificus infection has been advocated by some as a means for improving patient outcomes (3,10,14). However, the proper timing for surgery is debatable: in the early stages of infection, patients may not be stable enough to be moved to the operating room, and patients may have little chance of surviving if surgery is delayed until a later stage of infection (3). Because V. vulnificus infections can be life threatening, it is critical for emergency/trauma department doctors, including surgeons who make decisions regarding surgery for necrotizing fasciitis, to have early knowledge of a patient’s prognosis.
Some clinical data are available regarding the prognostic factors for patients with fatal V. vulnificus infection (10,15–17). Higher Acute Physiology and Chronic Health Evaluation (APACHE) II and Mortality in Emergency Department Sepsis (MEDS) scores have been reported to be significant prognostic indicators for V. vulnificus–infected patients (10,15). However, the APACHE II and MEDS scoring systems might be too complex to use quickly in an emergency setting. Thus, to find a marker to predict the risk for death among V. vulnificus–infected patients, we investigated the initial clinical and laboratory data available in hospital emergency departments. The aims of our study were to describe the clinical outcomes of V. vulnificus–infected patients and identify an accurate and simple predictive marker for death.
Patients
For the study, we identified 34 V. vulnificus–infected patients (>18 years of age) who had been admitted during January 2000–December 2011 to Chosun University Hospital, a tertiary teaching hospital in Gwangju, South Korea. V. vulnificus infection was diagnosed on the basis of blood and/or wound culture results. Clinical isolates were identified by using a Vitek II automated system (bioMérieux, Marcy l’Étoile, France). The study was approved by the Ethics in Human Research Committee of Chosun University Hospital.
Data Collection and Definitions
Demographic information and data regarding patients’ clinical manifestations, laboratory variables, operations, antimicrobial drug regimens, underlying diseases, and time to death were obtained by chart review. In Chosun University Hospital, arterial blood gas analysis and blood cultures are routinely performed before starting empirical antimicrobial drug treatment in patients with signs or symptoms of infection.
The use of an appropriate antimicrobial drug regimen was noted if a patient had been administered ceftriaxone, cefotaxime, or ciprofloxacin alone or in combination with doxycycline. Chronic liver disease was noted if a patient had shown clinical or laboratory signs of chronic liver disease caused by alcoholism or by hepatitis B or C virus. Fasciotomy was noted if surgery had been performed for necrotizing fasciitis. Death was defined as dying while in the hospital.
Statistical Analyses
We compared demographic and clinical characteristics for patients who survived and those who did not by using t-tests for continuous variables and χ2 tests for categorical variables. For APACHE II scores and pH levels at admission, we calculated the areas under the receiver operating characteristic curves (AUROCs) for predicting death caused by V. vulnificus infection. Sensitivities, specificities, positive predictive values, negative predictive values, positive likelihood ratios (how much to increase the probability of survival if the test is positive), and negative likelihood ratios (how much to decrease the probability of survival if the test is negative) of various APACHE II scores and pH cutoff levels on admission were calculated (18). The Youden index was used to determine the optimal cutoff value (19).
Multivariate analyses were performed by using the Cox proportional hazards model; variables that showed a p value of <0.05 in univariate analyses were included in these analyses. Of the variables for arterial blood gas analysis, pH was the only variable included in the model, and the APACHE II score was excluded from the multivariate model because of collinearity with pH and creatinine. Survival curves were constructed by using the Kaplan–Meier method, and a log-rank test was used for comparison. Survival was measured from the time of hospital admission to the time of death. We evaluated death status as death occurring <120 h after admission; patients who lived >120 h after admission were not included in the death statistics. A 2-tailed p value of <0.05 was considered to indicate statistical significance. All statistical analyses were performed by using SAS software, version 8.2 (SAS Institute Inc., Cary, NC, USA).
Clinical Outcomes for Patients with V. vulnificus Infection
Of the 34 patients with a diagnosis of V. vulnificus infection, 3 (9%) were female and 31 (91%) were male. A total of 25 patients had a history of consuming raw seafood, 5 had a wound exposure to seawater, and 4 were unaware of their exposure source. Of the 32 patients who had chronic liver disease, 27 (84%) had chronic alcoholism and 5 (16%) had hepatitis B virus infection.Of the 34 total patients, 16 (47%) died and 18 (53%) survived. The median time between arrival at the hospital and death was 15 h (range 4–70 h). For patients who survived and those who did not, the differences in underlying diseases, interval between symptom onset and hospital admission, initial appropriate antimicrobial drug treatment, and surgical treatment were not significant (Table 1).
Predictive Markers for Death
Univariate analysis showed that activated partial thromboplastin time and levels of pH, partial pressure of oxygen, serum bicarbonate, blood urea nitrogen, creatinine, aspartate transaminase, and alanine transaminase were significantly different between patients who survived and those who did not survive (Table 2). In multivariate analysis, initial pH on arterial blood gas analysis and serum creatinine level were significant risk factors for death (Table 3).
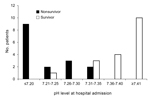
We found a significant difference in survival for patients with different arterial pH levels at hospital admission (Figure 1). No patients with a pH level of <7.2 at admission survived; all patients with a pH level of >7.35 survived (Figure 1, 2). An optimal pH cutoff level of <7.35 had a sensitivity of 100%, specificity of 83%, positive predictive value of 84%, negative predictive value of 100%, positive likelihood ratio of 6.0, and negative likelihood ratio of 0. An optimal APACHE II cutoff score of >14 had a sensitivity of 75%, specificity of 67%, positive predictive value of 67%, negative predictive value of 75%, positive likelihood ratio of 2.3, and negative likelihood ratio of 0.3 (Table 4). The AUROC of pH for prediction of death was 0.972 (95% CI 0.924–1.000), and the AUROC of the APACHE II score was 0.746 (95% CI 0.595–0.933) (p = 0.005) (Figure 3).
In this study, approximately half of the patients with V. vulnificus infection died, and death occurred <72 h after hospital admission. Patient survival differed significantly by pH level at hospital admission, and the initial pH level for patients was a more accurate predictive marker for death than was the APACHE II score.
Many studies have asserted that antimicrobial drugs should be immediately administered to patients with suspected V. vulnificus infection (3,8,15,20). Other studies suggest that a combination of antimicrobial drugs (i.e., third-generation cephalosporin, ciprofloxacin, and doxycycline) is the best treatment for V. vulnificus infection (21,22). In addition, surgical intervention is necessary to remove necrotic tissue and bacteria (20). Survival of patients with necrotizing fasciitis has been shown to improve when adequate debridement and fasciotomy are performed early in infection (3,20,23,24).
However, in our study there was no difference in the time from symptom onset to hospital admission and the timing of the initiation of appropriate antimicrobial drug treatment and surgical treatment for patients who survived and those who did not. This lack of difference may partly be due to the fact that patients died too early after admission to benefit from antimicrobial drug treatment or surgery. The median time to death for nonsurviving patients in our study was 15 h, even though they arrived at the hospital relatively early after symptom onset. The time to death in our study seems to be shorter than that in another study, which showed 11 (73%) of the 15 deaths occurred <72 h after arrival at the hospital (15).
That earlier study and another study reported that APACHE II and MEDS scores at hospital admission were useful prognostic indicators in primary septicemia or wound infections caused by V. vulnificus (10,15). In another study, the level of V. vulnificus DNA was substantially higher in nonsurvivors than in survivors of V. vulnificus septicemia, and the V. vulnificus DNA load correlated with the APACHE II score (17). Our data also showed that the APACHE II score may be useful for prediction of death in V. vulnificus–infected patients, although the AUROC for the APACHE II score was significantly less than that for initial pH.
In previous reports, hemorrhagic bullae formation, necrotizing fasciitis, and septic shock were risk factors for death in patients with V. vulnificus infection (8,25–27). Although in our study, no skin or soft tissue lesions were associated with death, the pH and creatinine levels at hospital admission were independently associated with death of V. vulnificus–infected patients. Low arterial pH level might result from lactic acidosis caused by marked tissue hypoperfusion in shock and damage to extremities by necrotizing fasciitis (28,29). The finding that high creatinine level was associated with death also supports the possibility that hypoperfusion or sepsis contributed to the death of patients in this study because azotemia might result from renal hypoperfusion or septic renal injury.
Our study did have some limitations because of its retrospective design and the relatively small number of study patients, which may hamper generalization of the results. In addition, because most patients in this study had chronic liver disease, the study findings might not be applicable to patients with various concurrent diseases other than chronic liver disease.
Despite these limitations, our study may have relevant clinical implications. Our data demonstrate that the pH level determined by arterial blood gas analysis at hospital admission is more accurate than the APACHE II score as a marker for predicting death in V. vulnificus–infected patients. Arterial blood gas levels are easy to determine, and results are quickly available; thus, this test is useful in emergency or trauma departments.
We propose that arterial pH level should be determined for patients with suspected V. vulnificus infection as soon as possible after hospital admission. When the arterial pH level is low (<7.2), patient outcome may be unsatisfactory, regardless of emergency surgery and appropriate antimicrobial drug therapy. When the pH level is not low, patient outcome may be excellent if the patient receives appropriate antimicrobial drug treatment and, if needed, surgery.
Dr. Yun is a medical doctor specializing in infectious disease and tropical medicine at the Medical School of Chosun University, Gwangju, South Korea. Her research interests focus on tropical diseases.
Acknowledgment
This study was supported by research funds (2011) from Chosun University.
References
- Chuang YC, Young CD, Chen CW. Vibrio vulnificus infection. Scand J Infect Dis. 1989;21:721–6. DOIPubMedGoogle Scholar
- Okoh AI, Sibanda T, Nongogo V, Adefisoye M, Olayemi OO, Nontongana N. Prevalence and characterisation of non-cholerae Vibrio spp. in final effluents of wastewater treatment facilities in two districts of the Eastern Cape Province of South Africa: implications for public health. Environ Sci Pollut Res Int. 2014 [cited 2014 Nov19]. Epub 2014 Aug 29.
- Huehn S, Eichhorn C, Urmersbach S, Breidenbach J, Bechlars S, Bier N, Pathogenic vibrios in environmental, seafood and clinical sources in Germany. Int J Med Microbiol. 2014;•••:S1438–4221.PubMedGoogle Scholar
- Lee YC, Hor LI, Chiu HY, Lee JW, Shieh SJ. Prognostic factor of mortality and its clinical implications in patients with necrotizing fasciitis caused by Vibrio vulnificus. Eur J Clin Microbiol Infect Dis. 2014;33:1011–8. DOIPubMedGoogle Scholar
- Hsueh PR, Lin CY, Tang HJ, Lee HC, Liu JW, Liu YC, Vibrio vulnificus in Taiwan. Emerg Infect Dis. 2004;10:1363–8. DOIPubMedGoogle Scholar
- Newton A, Kendall M, Vugia DJ, Henao OL, Mahon BE. Increasing rates of vibriosis in the United States, 1996–2010: review of surveillance data from 2 systems. Clin Infect Dis. 2012;54(Suppl 5):S391–5. DOIPubMedGoogle Scholar
- Chuang YC, Yuan CY, Liu CY, Lan CK, Huang AH. Vibrio vulnificus infection in Taiwan: report of 28 cases and review of clinical manifestations and treatment. Clin Infect Dis. 1992;15:271–6. DOIPubMedGoogle Scholar
- Howard RJ, Bennett NT. Infections caused by halophilic marine Vibrio bacteria. Ann Surg. 1993;217:525–31. DOIPubMedGoogle Scholar
- Liu JW, Lee IK, Tang HJ, Ko WC, Lee HC, Liu YC, Prognostic factors and antibiotics in Vibrio vulnificus septicemia. Arch Intern Med. 2006;166:2117–23. DOIPubMedGoogle Scholar
- KuoChou TN. Chao WN, Yang C, Wong RH, Ueng KC, Chen SC. Predictors of mortality in skin and soft-tissue infections caused by Vibrio vulnificus. World J Surg. 2010;34:1669–75. DOIPubMedGoogle Scholar
- Chen SC, Chan KS, Chao WN, Wang PH, Lin DB, Ueng KC, Clinical outcomes and prognostic factors for patients with Vibrio vulnificus infections requiring intensive care: a 10-yr retrospective study. Crit Care Med. 2010;38:1984–90 .PubMedGoogle Scholar
- Tsai YH, Hsu RW, Huang KC, Huang TJ. Laboratory indicators for early detection and surgical treatment of vibrio necrotizing fasciitis. Clin Orthop Relat Res. 2010;468:2230–7. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Vibrio vulnificus infections associated with raw oyster consumption—Florida, 1981–1992. MMWR Morb Mortal Wkly Rep. 1993;42:405–7 .PubMedGoogle Scholar
- Neupane GP, Kim DM. In vitro time-kill activities of ciprofloxacin alone and in combination with the iron chelator deferasirox against Vibrio vulnificus. Eur J Clin Microbiol Infect Dis. 2010;29:407–10. DOIPubMedGoogle Scholar
- Halow KD, Harner RC, Fontenelle LJ. Primary skin infections secondary to Vibrio vulnificus: the role of operative intervention. J Am Coll Surg. 1996;183:329–34 .PubMedGoogle Scholar
- Chou TN, Lee YT, Lai YY, Chao WN, Yang C, Chen CC, Prognostic factors for primary septicemia and wound infection caused by Vibrio vulnificus. Am J Emerg Med. 2010;28:424–31. DOIPubMedGoogle Scholar
- Hou CC, Lai CC, Liu WL, Chao CM, Chiu YH, Hsueh PR. Clinical manifestation and prognostic factors of non-cholerae Vibrio infections. Eur J Clin Microbiol Infect Dis. 2011;30:819–24. DOIPubMedGoogle Scholar
- Kim DM, Jung SI, Jang HC, Lee CS, Lee H, Yun NR, Vibrio vulnificus DNA load and mortality. J Clin Microbiol. 2011;49:413–5. DOIPubMedGoogle Scholar
- Horseman MA, Surani S. A comprehensive review of Vibrio vulnificus: an important cause of severe sepsis and skin and soft-tissue infection. Int J Infect Dis. 2011;15:e157–66. DOIPubMedGoogle Scholar
- Chen SC, Lee YT, Tsai SJ, Chan KS, Chao WN, Wang PH. etal. Antibiotic therapy for necrotizing fasciitis caused by Vibrio vulnificus: retrospective analysis of an 8 year period. J Antimicrob Chemother. 2012;67:488–93. DOIPubMedGoogle Scholar
- Jang HC, Choi SM, Kim HK, Kim SE, Kang SJ, Park KH. etal. Invivo efficacy of the combination of ciprofloxacin and cefotaxime against Vibrio vulnificus sepsis. PLoS ONE. 2014;9:e101118. DOIPubMedGoogle Scholar
- Kuo YL, Shieh SJ, Chiu HY, Lee JW. Necrotizing fasciitis caused by Vibrio vulnificus: epidemiology, clinical findings, treatment and prevention. Eur J Clin Microbiol Infect Dis. 2007;26:785–92 . DOIPubMedGoogle Scholar
- Chao WN, Tsai CF, Chang HR, Chan KS, Su CH, Lee T, Impact of timing of surgery on outcome of Vibrio vulnificus-related necrotizing fasciitis. Am J Surg. 2013;206:32–9. DOIPubMedGoogle Scholar
- Klontz KC, Lieb S, Schreiber M, Janowski HT, Baldy LM, Gunn RA. Syndromes of Vibrio vulnificus infections. Clinical and epidemiologic features in Florida cases, 1981–1987. Ann Intern Med. 1988;109:318–23. DOIPubMedGoogle Scholar
- Chung PH, Chuang SK, Tsang T, Wai-man L, Yung R, Lo J. Cutaneous injury and Vibrio vulnificus infection. Emerg Infect Dis. 2006;12:1302–3. DOIPubMedGoogle Scholar
- Penman AD, Lanier DC Jr, Avara WT III, Canant KE, DeGroote JW, Brackin BT, Vibrio vulnificus wound infections from the Mississippi Gulf coastal waters: June to August 1993. South Med J. 1995;88:531–3 . DOIPubMedGoogle Scholar
- Mikkelsen ME, Miltiades AN, Gaieski DF, Goyal M, Fuchs BD, Shah CV, Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit Care Med. 2009;37:1670–7 . DOIPubMedGoogle Scholar
Figures
Tables
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to http://www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title:
pH Level as a Marker for Predicting Death among Patients with Vibrio vulnificus Infection, South Korea, 2000–2011
CME Questions
1. You are seeing a 50-year-old man with a 2-day history of vomiting and fever. Blistering skin lesions have now developed in this patient, and you suspect that he might have an infection with Vibrio vulnificus. Which of the following statements characterizes the cohort of patients with V. vulnificus in the current study?
A. The cohort was equally divided between women and men
B. Less than 10% of patients had a history of eating raw seafood
C. The majority of patients had a history of potential exposure to V. vulnificus
D. Chronic liver disease was rare overall
2. Which of the following statements regarding outcomes of patients with V. vulnificus infection in the current study is most accurate?
A. Nearly half of patients died
B. The median time between hospital arrival and time of death was 75 hours
C. Initial application of appropriate antibiotics dramatically improved survival
D. Early surgery dramatically improved survival
3. You want a quick measure of prognosis for this critically ill patient. Which of the following variables was most significantly associated with survival from infection with V. vulnificus on multivariate analysis in the current study?
A. Partial pressure of oxygen
B. Serum creatinine
C. Serum transaminase levels
D. Active partial thromboplastin time
4. The patient’s ABGA results return. What was the optimal cutoff value for pH on ABGA in predicting mortality in the current study?
A. 7.0
B. 7.15
C. 7.35
D. 7.8
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
Related Links
Table of Contents – Volume 21, Number 2—February 2015
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Dong-Min Kim, Chosun University School of Medicine, 588 seo-suk dong, Dong gu, Gwangju city, Republic of Korea, 501-717
Top