Volume 25, Number 6—June 2019
Research
Performance of 2 Commercial Serologic Tests for Diagnosing Zika Virus Infection
Figure 2
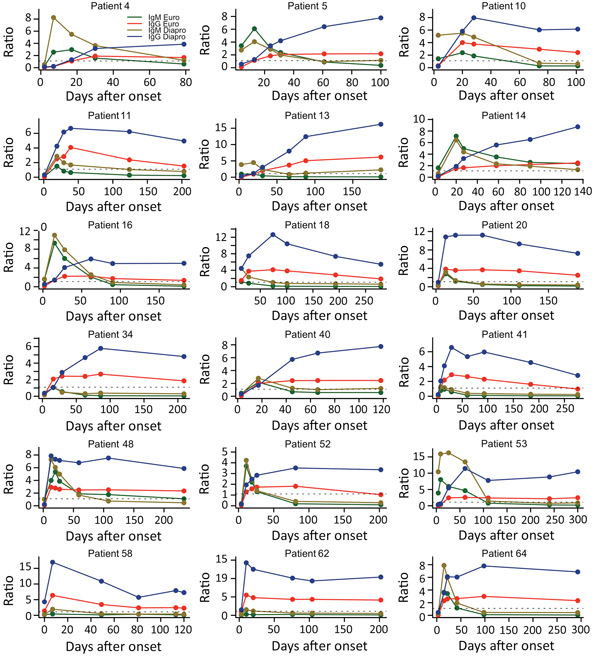
Figure 2. Individual time-course analyses of Zika virus IgM and IgG signal-to-cutoff ratios obtained by using Euroimmun and Dia.Pro kits for 18 patients for whom 5 or more sequential samples were available.
Page created: May 20, 2019
Page updated: May 20, 2019
Page reviewed: May 20, 2019
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.