Volume 3, Number 2—June 1997
Perspective
The Economic Impact of a Bioterrorist Attack: Are Prevention and Postattack Intervention Programs Justifiable?
Figure 2
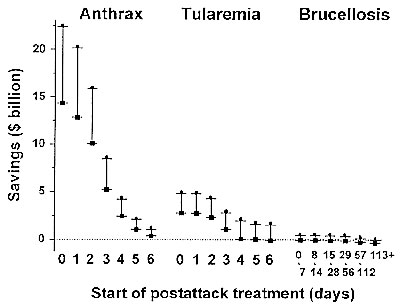
Figure 2. Rangesa of net savings due to postattack prophylaxis by disease and day of prophylaxis program initiation. aMaximum savings (l) were calculated by assuming a 95% effectiveness prophylaxis regimen and a 3% discount rate in determining the present value of expected lifetime earnings lost due to premature death (16) and a multiplication factor of 5 to adjust for unnecessary prophylaxis. Minimum savings (n) were calculated by assuming an 80% to 90% effectiveness regimen and a 5% discount rate and a multiplication factor of 15. In tularemia prophylaxis programs initiated on days 4-7 postattack, the minimum savings were calculated by assuming a 95% prophylaxis regimen effectiveness rather than an effectiveness of 80% to 90%.
References
- Abramova FAN, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993;90:2291–4. DOIPubMedGoogle Scholar
- Benenson AS, ed. Control of communicable diseases manual. 16th ed. Washington (DC): American Public Health Association, 1995.
- Messelson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, The Sverdlosvsk anthrax outbreak of 1979. Science. 1994;266:1202–8. DOIPubMedGoogle Scholar
- Kaufmann AF, Fox MD, Boyce JM, Anderson DC, Potter ME, Martone WJ, Airborne spread of brucellosis. Ann N Y Acad Sci. 1980;335:105–14. DOIGoogle Scholar
- Olle-Goig JE, Canela-Soler J. An outbreak of Brucella melitensis infection by airborne transmission among laboratory workers. Am J Public Health. 1987;77:335–8. DOIPubMedGoogle Scholar
- Staszkiewicz J, Lewis CM, Colville J, Zervos M, Band J. Outbreak of Brucella melitensis among microbiology laboratory workers in a community hospital.J Clin Microbiol. 1991;29:287–90. PubMedGoogle Scholar
- Trever RW, Cluff LE, Peeler RN, Bennett IL. Brucellosis I. laboratory-acquired acute infection. Arch Intern Med. 1959;103:381–97.
- McCrumb FR. Aerosol infection of man with Pasteurella tularensis. Bacteriol Rev. 1961;25:262–7.PubMedGoogle Scholar
- Saslaw S, Eigelsbach HT, Wilson HR, Prior JA, Carhart S. Tularemia vaccine study II. respiratory challenge. Arch Intern Med. 1961;107:689–701.PubMedGoogle Scholar
- Friedlander AM, Welkos SL, Pitt MLM, Ezzell JW, Worsham PL, Rose KJ, Postexposure prophylaxis against experimental inhalation anthraxJ Infect Dis. 1993;167:1239–42.PubMedGoogle Scholar
- Sawyer WD, Dangerfield HG, Hogge AL, Crozier D. Antibiotic prophylaxis and therapy of airborne tularemia PubMed. Bacteriol Rev. 1966;30:542–8.PubMedGoogle Scholar
- Solera J, Rodriguez-Zapata M, Geijo P, Largo J, Paulino J, Saez L, Doxycycline-rifampin versus doxycycline-streptomycin in treatment of human brucellosis due to Brucella melitensis.Antimicrob Agents Chemother. 1995;39:2061–7. PubMedGoogle Scholar
- Luce BR, Manning WG, Siegel JE, Lipscomb J. Estimating costs in cost-effectiveness analysis. In: Gold MR, Siegel JE, Russell LB, Weinstein MC, editors. Cost-effectiveness in health and medicine. New York: Oxford University Press, 1966:176-213.
- Haddix AC, Teutsch SM, Shaffer PA, Dunet DO, eds. Prevention effectiveness: a guide to decision analysis and economic evaluation. New York: Oxford University Press, 1996.
- Lipscomb J, Weinstein MC, Torrance GW. Time preference. In: Gold MR, Siegel JE, Russell LB, Weinstein MC, editors. Cost-effectiveness in health and medicine. New York: Oxford University Press, 1966:214-35.
- U.S. Bureau of the Census. Statistical abstract of the United States: 1995. 115th ed. Washington (DC): U.S. Government Printing Office, 1996.
- National Center for Health Statistics. Health, United States, 1995. Hyattsville (MD):U.S.Departmentof Health and Human Services, Public Health Service, 1996.
- HealthCare Consultants of America, Inc. HealthCare Consultants' 1996 physicians fee and coding guide. 6th ed. Augusta (GA): HealthCare Consultants of America, Inc. 1996.
- Cardinale V, ed. 1996 Drug Topics Red Book. Montvale (NJ): Medical Economics Company, Inc., 1996.
- Robison LJ, Barry PJ. The competitive firm's response to risk. New York: Macmillan, 1987.
Page created: December 21, 2010
Page updated: December 21, 2010
Page reviewed: December 21, 2010
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.