Volume 4, Number 2—June 1998
Dispatch
Dual Infection with Ehrlichia chaffeensis and a Spotted Fever Group Rickettsia: A Case Report
Abstract
Well-documented cases of simultaneous human infection with more than one tick-borne pathogen are rare. To our knowledge only two dual infections have been reported: simultaneous human infection with the agent of human granulocytic ehrlichiosis and Borrelia burgdorferi and simultaneous human infection with B. burgdorferi and Babesia microti (1-2). Rocky Mountain spotted fever has long been known to be endemic in North Carolina; cases of human ehrlichial infection were recognized there soon after Ehrlichia chaffeensis was recognized as an important cause of tick-borne disease in the southeastern United States. Because both Rocky Mountain spotted fever and ehrlichiosis are prevalent in North Carolina, occasional cases of simultaneous human infection by rickettsial and ehrlichial agents would not be surprising; however, no such cases seem to have been reported.
A 44-year-old man with a history of hepatitis C infection and regular use of cocaine was asymptomatic until June 6, 1995, when he noted the acute onset of myalgia, headache, arthralgia, fever (40°C), nonproductive cough, nausea, and vomiting. The following day he sought medical care and received oral trimethoprim/sulfamethoxazole for a presumed respiratory tract infection. Over the next 3 days, his symptoms worsened; on the fourth day he noticed a maculopapular rash and came to our medical center. Initial examination showed tachycardia (pulse 125 beats per minute), fever (38.6°C), and a macular red rash localized to his right arm. The patient reported a tick bite 1 day before his symptoms began. In addition, he reported removing an engorged tick from his pet dog and crushing it between his fingers 4 days before he became ill. Laboratory studies showed leukopenia (white blood cell count 2,700/mm3), thrombocytopenia (platelet count 69,000/mm3), hyponatremia (serum sodium 130 meq/dL), and mildly elevated serum concentrations of hepatic enzymes (serum aspartate aminotransferase level of 160 IU/L [normal range 10 to 60 IU]). A screening test for HIV infection was negative. Therapy with intravenous doxycycline was begun (100 mg twice daily). A skin biopsy showed a perivascular lymphocytic infiltrate in the superficial and deep dermis. Direct immunofluorescent antibody staining of the fresh frozen skin biopsy with antiserum against Rickettsia rickettsii demonstrated organisms typical of spotted fever group rickettsiae within endothelial cells. Sections were also stained with antiserum against human albumen as a negative control. Parallel staining of a section of positive control tissue was also performed (3). Three days after admission, the patient was afebrile and asymptomatic. He was discharged and instructed to continue therapy with oral doxycycline for an additional 7 days. At follow-up 12 weeks later, he felt well. His white blood count was 7,600/mm3, and his platelet count was 166,000/mm3.
Serum samples obtained 5, 24, and 55 days after onset of illness were tested for antibodies to Ehrlichia chaffeensis and R. rickettsii antigens by immunofluorescence assay (IFA) except that E. chaffeensis (Arkansas strain) was used instead of E. canis (4,5).
A fourfold IFA antibody rise (1:32 to 1:128) occurred against E. chaffeensis antigen, but antibodies against R. rickettsii antigen were not detected in either the acute- or first convalescent-phase serum sample. However, a serum sample collected 55 days after onset contained an antibody titer of 1:40 to R. rickettsii antigen. Significant in vitro T-cell proliferation was seen in response to the antigens of both E. chaffeensis and R. rickettsii (Table 1).
A sample of blood taken before the patient was treated with doxycycline was centrifuged in two steps to remove red blood cells and to pellet buffy coat cells. A portion of cellular material was then resuspended in plasma and injected into Vero cells and 030 cells (North Carolina State University canine monocytoid cell line). Vero cell cultures were then maintained with medium 199 and 030 cells with RPMI 1640 containing 10% fetal bovine serum, 2 mM L-glutamine, and 0.075% sodium bicarbonate. Cultures were examined weekly for rickettsia-like organisms (5) or morulaelike structures (6) by light microscopy. Cell suspensions were stained by Gimenez, Wright, and direct immunofluorescent antibody (DFA) for R. rickettsii (rabbit anti-R. rickettsii, Centers for Disease Control and Prevention [CDC], Atlanta, GA) and E. chaffeensis (rabbit anti-ehrlichia fluorescein isothiocyanate-labeled conjugates, J.E. Dawson, CDC, Atlanta, GA) (6,7).
Approximately 12 weeks after onset of illness, venous blood was obtained and placed in tubes with EDTA anticoagulant. The specimen was shipped overnight on wet ice and then processed as follows. An equal volume of medium was added to a 10-ml aliquot of the blood sample, loaded onto 6 ml of Ficoll-paque (Pharmacia, Uppsala, Sweden), and centrifuged at 770 x g for 20 min at room temperature. The upper layer of plasma was removed. Then the interface (containing most of the lymphocytes) and the Ficoll-paque (containing most of the monocytes) were harvested and washed once with medium. The pellet was suspended in medium (Dulbecco modified Eagle medium [DMEM]) containing 10% fetal bovine serum, 2 mM glutamine, 5 x 10-5 M 2-mercaptoethanol, 10 µM HEPES buffer, and 50 µg/ml of gentamicin. The concentration of cells was adjusted to 1 x 106/ml in DMEM. A 0.1-ml portion of these cells was then aliquoted into each well of a 96-well plate. Serial dilutions of inactivated intact rickettsial or ehrlichial antigen (0.2µg to 4 µg/well) were added to individual wells in the same plate in triplicate. The plate was then incubated at 37°C in a 5% CO2 atmosphere for 6 days. Afterwards, 3H-thymidine was added to each well (1 microcurie/well), and the plates were again incubated at 37°C overnight. On the next day the cells were harvested and dried; ß-fluor cocktail scintillation fluid was then added, and the mixture was counted in a ß-counter. Count per minute rates greater than threefold of the unstimulated control were considered positive.
After 8 days of incubation in Vero cells, a spotted fever group rickettsia was detected by direct immunofluorescent antibody from a sample of blood obtained immediately before doxycycline therapy. However, the rickettsial organism did not thrive with subsequent in vitro passage, and further attempts to recover the organism in vitro or by injecting into guinea pigs were unsuccessful. Subsequent attempts to amplify rickettsial 17kDa protein gene and the 16SrRNA gene by PCR with stored cell culture materials from passages 1 and 2 were negative. In addition, morulae consistent with ehrlichial organisms were detected by light and direct fluorescence in blood taken from the patient before antimicrobial therapy. Subsequent attempts to isolate ehrlichiae with further passage in 030 cells were unsuccessful.
DNA was extracted from acute-phase blood with a commercially available kit (IsoQuick; Orca Research Inc., Bothell, WA) for preparation of DNA from whole blood, according to manufacturer's instructions. The dried pellet was then resuspended in DNAase-free water in preparation for PCR analysis.
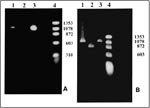
A pair of primers that recognize sequences of the nadA gene of E. chaffeensis was used to analyze the patient's sample (Table 2). The PCR products were then separated by electrophoresis in a 1% agarose gel, stained with ethidium bromide, and visualized under UV light (Figure A).
The amplification conditions for the 120 kDA protein gene were essentially the same as the ones used for the nadA gene, except that nested PCR was performed (9-10). The outside primers were pXCF3 and pXAR4, and the nested primers were pXCF3b and pXAR5 (Table 2). The nested primers obtained a product similar to the 1.1 kb product amplified from the Arkansas strain and one repeat unit larger than the Sapulpa strain (Figure B).
Nested PCR amplification of the 120 kDa protein gene of E. chaffeensis yielded a PCR product from the patient's blood similar to the repeat region of the E. chaffeensis Arkansas strain. The nadA gene of E. chaffeensis was amplified by a single pair of primers; the sequence of the nadA segment was determined to be similar in size to that of E. chaffeensis (Figure).
This patient had clinical features of infection with both R. rickettsii and E. chaffeensis. His rash, although limited and purely macular, was compatible with R. rickettsii infection (11). Compelling data support the conclusion that the patient was infected with a spotted fever group rickettsia: 1) IFA staining of a skin specimen showed organisms typical of spotted fever group rickettsia, 2) a spotted fever group rickettsia was initially recovered in tissue culture from a blood sample taken before therapy, and 3) T-cell lymphocytes harvested from a convalescent-phase blood sample contained specific memory cells reactive to R. rickettsii from which a T- lymphocyte cell line was subsequently established.
We do not know why a typical vigorous convalescent-antibody response to R. rickettsii did not develop in this patient, even though antibody response to E. chaffeensis was typical. It is possible, but highly unlikely, that the spotted fever group rickettsia isolated from the patient was a species other than R. rickettsii. We cannot state with certainty that the isolate detected in vitro in Vero cells and then lost in subsequent passage was indeed R. rickettsii and not a closely related species such as R. amblyommii (12). It is also possible that coinfection with E. chaffeensis suppressed a normal antibody response to R. rickettsii antigens. Alternately, if infection with E. chaffeensis was transmitted by a tick bite several days before the second bite by a tick infected with a spotted fever group rickettsia, antimicrobial therapy may have blunted a typical antibody response to rickettsial—but not to previously present ehrlichial—antigens (early antimicrobial therapy can impair or blunt the antibody response in patients with R. rickettsii infection [13]). In addition, a previous report described a patient with documented rickettsemia who was treated within 12 hours of onset of symptoms and failed to develop a convalescent-phase antibody response (14).
Had we not performed a skin biopsy or attempted to isolate rickettsiae from the blood, we might have logically (but erroneously) concluded that the illness was due solely to E. chaffeensis infection.
Concurrent infection with E. chaffeensis is supported by three different lines of evidence: 1) a greater than fourfold rise in IFA titer against E. chaffeensis antigens, 2) in vitro stimulation of T cells harvested from a convalescent-phase blood sample with the antigen of E. chaffeensis causing a strong lymphoproliferative response, and 3) PCR products similar in size to the repeat region of the 120 kDA surface protein gene of E. chaffeensis Arkansas strain and specific for the E. chaffeensis nadA gene recovered from a sample of blood obtained before therapy. Although we did not sequence amplicon generated from the 120 kDa protein gene, the oligonucleotide primers that were used in PCR amplification are derived from regions of the 120 kDa protein gene that are unique to E. chaffeensis. These regions are not present in other bacteria, including closely related Ehrlichia species.
We are unable to determine if the patient was infected by two different ticks or one tick simultaneously infected with E. chaffeensis and R. rickettsii or another spotted fever group rickettsia. Coinfection of Ixodes scapularis ticks with the agent of human granulocytic ehrlichiosis or a closely related organism and B. burgdorferi has been described (15-17); however, to our knowledge, simultaneous infection of a tick with R. rickettsii and E. chaffeensis has not. Furthermore, the major vector of E. chaffeensis in North Carolina is thought to be Amblyomma americanum (18), and the primary vector for R. rickettsii in this area is Dermacentor variabilis. Although E. chaffeensis has been detected on one occasion in D. variabilis (19), the evidence that A. americanum is a vector for Rocky Mountain spotted fever elsewhere is circumstantial; to our knowledge, R. rickettsii has never been isolated from A. americanum (W. Burgdorfer, pers. comm.).
Most likely E. chaffeensis was transmitted to the patient from A. americanum, and the spotted fever rickettsia was transmitted from D. variabilis. Indeed, a walk in the fields of the Piedmont region of North Carolina in the spring frequently results in attachment of numerous ticks of both species. Since the patient had direct contact or bites with two different ticks before his illness, we suspect that he was infected by separate ticks rather than by one tick infected with both pathogens.
Finally, this case is remarkable in an additional way. Despite coinfection with two pathogens, the patient had a relatively mild illness. He did not have shock, major organ dysfunction, or central nervous system involvement; and with doxycycline therapy, recovery was prompt and uneventful.
On the basis of this case report, we cannot predict that future cases will have a benign course or an impaired or absent convalescent-phase antibody response to one or more of the infecting agents. However, the patient's illness illustrates that coinfection with a spotted fever group rickettsia and E. chaffeensis may occur in areas where both diseases are endemic. The frequency of human coinfection with ehrlichial and rickettsial agents is unknown. Serologic studies have demonstrated that approximately one third of patients with antibodies to E. chaffeensis also have antibodies to one or more rickettsiae (20). Whether this phenomenon is due to the presence of cross-reactive antigens or actual immune stimulation by simultaneous or sequential multiple tick-borne pathogens is unknown.
Dr. Sexton is a professor in the Department of Medicine, Division of Infectious Diseases, Duke University Medical Center, Durham, North Carolina. His research interests include the clinical and epidemiologic features of Rocky Mountain spotted fever and other tick-borne diseases.
References
- Nadelman RB, Horowitz HW, Hsieh T-C, Wu JM, Aguero-Rosenfeld ME, Schwartz I, Simultaneous human granulocytic ehrlichiosis and Lyme borreliosis. N Engl J Med. 1997;337:27–30. DOIPubMedGoogle Scholar
- Krause PJ, Telford SR III, Spielman A, Sikand V, Ryan R, Christianson D, Concurrent Lyme disease and babesiosis: evidence for increased severity and duration of illness. JAMA. 1996;275:1657–60. DOIPubMedGoogle Scholar
- Procop GW, Burchette JL, Howell DN, Sexton DJ. Immunoperoxidase and immunofluorescent staining of Rickettsia rickettsii in skin biopsy. A comparative study. Arch Pathol Lab Med. 1997;121:894–9.PubMedGoogle Scholar
- Philip RN, Casper EA, Ormsbee RA, Peacock MG, Burgdorfer W. Microimmunofluorescence test for the serological study of Rocky Mountain spotted fever and typhus. J Clin Microbiol. 1976;3:51–61.PubMedGoogle Scholar
- Dawson JE, Rikihisa Y, Ewing SA, Fishbein DB. Serologic diagnosis of human ehrlichiosis using two Ehrlichia canis isolates. J Infect Dis. 1991;163:564–7.PubMedGoogle Scholar
- Lange JV, Walker DH, Wester TB. Documented Rocky Mountain spotted fever in wintertime. JAMA. 1982;247:2403–4. DOIPubMedGoogle Scholar
- Iqbal Z, Rikihisa Y. Re-isolation of Ehrlichia canis from blood and tissue of dogs after doxycyline treatment. J Clin Microbiol. 1994;32:1644–9.PubMedGoogle Scholar
- Xu X-J, Walker DH. Sequence and characterization of an Ehrlichia chaffeensis gene encoding 314 amino acids highly homologous to the NAD A enzyme. FEMS Microbiol Lett. 1997;154:53–8. DOIPubMedGoogle Scholar
- Xu X-J, Crocquet-Valdes PA, Walker DH. Cloning and sequencing of the gene for 120-kDA immunodominant surface protein of Ehrlichia chaffeensis. Gene. 1997;184:149–54. DOIPubMedGoogle Scholar
- Xu X-J, Piesman JF, Olson JG, Walker DH. Geographic distribution of different genetic types of Ehrlichia chaffeensis. Am J Trop Med Hyg. 1997;56:679–80.PubMedGoogle Scholar
- Sexton DJ, Corey GR. Rocky Mountain spotless and almost spotless fever: a wolf in sheep's clothing. Clin Infect Dis. 1992;15:439–48.PubMedGoogle Scholar
- Anderson BE, Sims KG, Olson JG, Childs JE, Piesman JF, Happ CM, Amblyomma americanum: a potential vector of human ehrlichiosis. Am J Trop Med Hyg. 1993;49:239–44.PubMedGoogle Scholar
- Schubert JH. Serologic titers in rickettsial infection as affected by antibiotic treatment. Pub Health Lab. 1952;10:38–41.
- Davis JP, Wilfert CM, Sexton DJ, Burgdorfer W, Casper EA, Philip RN. Serologic comparison of R. rickettsii isolated from patients in Montana and North Carolina. In: Rickettsiae and rickettsial diseases. New York: Academic Press, Inc.; 1981 p. 139-47.
- Pancholi P, Kolbert CP, Mitchell PD, Reed KD Jr, Dumler JS, Bakken JS, Ixodes dammini as a potential vector of human granulocytic ehrlichiosis. J Infect Dis. 1995;172:1007–12.PubMedGoogle Scholar
- Telford SR III, Dawson JE, Katavolos P, Warner CK, Kolbert CP, Persing DH. Perpetuation of the agent of human granulocytic ehrlichiosis in a deer tick-rodent cycle. Proc Natl Acad Sci U S A. 1996;93:6209–14. DOIPubMedGoogle Scholar
- Schwartz I, Fish D, Daniels TJ. Prevalence of the rickettsial agent of human granulocytic ehrlichiosis in ticks from a hyperendemic focus of Lyme disease. N Engl J Med. 1997;336:49–50. DOIGoogle Scholar
- Ewing SA, Dawson JE, Kocan AA, Barker RW, Warner CK, Panciera RJ, Experimental transmission of Ehrlichia chaffeensis (Rickettsiales: Ehrlichieae) among white-tailed deer by Amblyomma americanum (Acarei: Ixodidae). J Med Entomol. 1995;32:368–74.PubMedGoogle Scholar
- Anderson BE, Sumner JW, Dawson JE, Tzianabos T, Greene CR, Olson JC, Detection of the etiologic agent of human ehrlichiosis by polymerase chain reaction. J Clin Microbiol. 1992;30:775–80.PubMedGoogle Scholar
- Dawson JE, Fishbein DB, Eng TR, Redus MA, Greene NR. Diagnosis of human ehrlichiosis with the indirect fluorescent antibody test: kinetics and specificity. J Infect Dis. 1990;162:91–5.PubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 4, Number 2—June 1998
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Daniel J. Sexton, Box 3605, Duke University Medical Center, Durham, NC 27710, USA; fax: 919-681-7945
Top